 
To compare Non-Urothelial Tract Recurrence Free Survival (NUTRFS) for nivolumab versus placebo in subjects with tumors expressing PD-L1 (>= 1% membranous staining in tumor cells) and all randomized subjects Sujetos con tumores que expresan PD-L1 (> =1% tinción membranosa en las células tumorales) y subjects with tumors expressing PD-L1 (>=1% membranous staining in tumor cells)Ĭomparar la supervivencia libre de enfermedad (SLE) con nivolumab frente a placebo en: To compare the disease free survival (DFS) for nivolumab versus placebo in: Sujetos que se han sometido a resección radical del carcinoma urotelial invasivo originado en la vejiga o en el tracto urinario superior ( pelvis renal o ureter) con un alto riesgo de recidivaĮ.1.2 Medical condition or disease under investigationĬondition being studied is a rare disease Subjects who have undergone radical resection of Invasive urothelial carcinoma originating in the bladder or upper urinary tract (renal pelvis or ureter) and are at high risk of recurrence. 
Medical condition in easily understood language 
Sujetos con Carcinoma urotelial invasivo de alto riesgo Subjects with High Risk Invasive Urothelial Carcinoma Medicinal product containing genetically modified organismsĮ.1 Medical condition or disease under investigation Immunological medicinal product (such as vaccine, allergen, immune serum) one involving a medical device)Ĭommittee on Advanced therapies (CAT) has issued a classification for this productĬombination product that includes a device, but does not involve an Advanced Therapy The IMP has been designated in this indication as an orphan drug in the Communityĭ.3.8 to D.3.10 IMP Identification Details (Active Substances)Īctive substance of biological/ biotechnological origin (other than Advanced Therapy IMP (ATIMP)Ĭombination ATIMP (i.e. IMP to be used in the trial has a marketing authorisation Status of the IMP to be used in the clinical trial Trial is part of a Paediatric Investigation PlanĮMA Decision number of Paediatric Investigation Plan WHO Universal Trial Reference Number (UTRN) Name or abbreviated title of the trial where available Nivolumab versus placebo after surgery in subjects with urinary tract cancerĮstudio de Nivolumab frente a placebo después de cirugía en sujetos con Carcinoma urotelial Title of the trial for lay people, in easily understood, i.e. Phase 1 trials conducted solely on adults and that are not part of an agreed paediatric investigation plan (PIP) are not publicly available (seeĪ Phase 3 Randomized, Double-blind, Multi-center Study of Adjuvant Nivolumab versus Placebo in Subjects with High Risk Invasive Urothelial CarcinomaĮstudio de fase 3 aleatorizado, doble ciego, multicéntrico, de Nivolumab como tratamiento adyuvante frente a placebo en sujetos con Carcinoma urotelial invasivo de alto riesgo Older paediatric trials (in scope of Article 45 of the Paediatric Regulation (EC) No 1901/2006). The register also displays information on 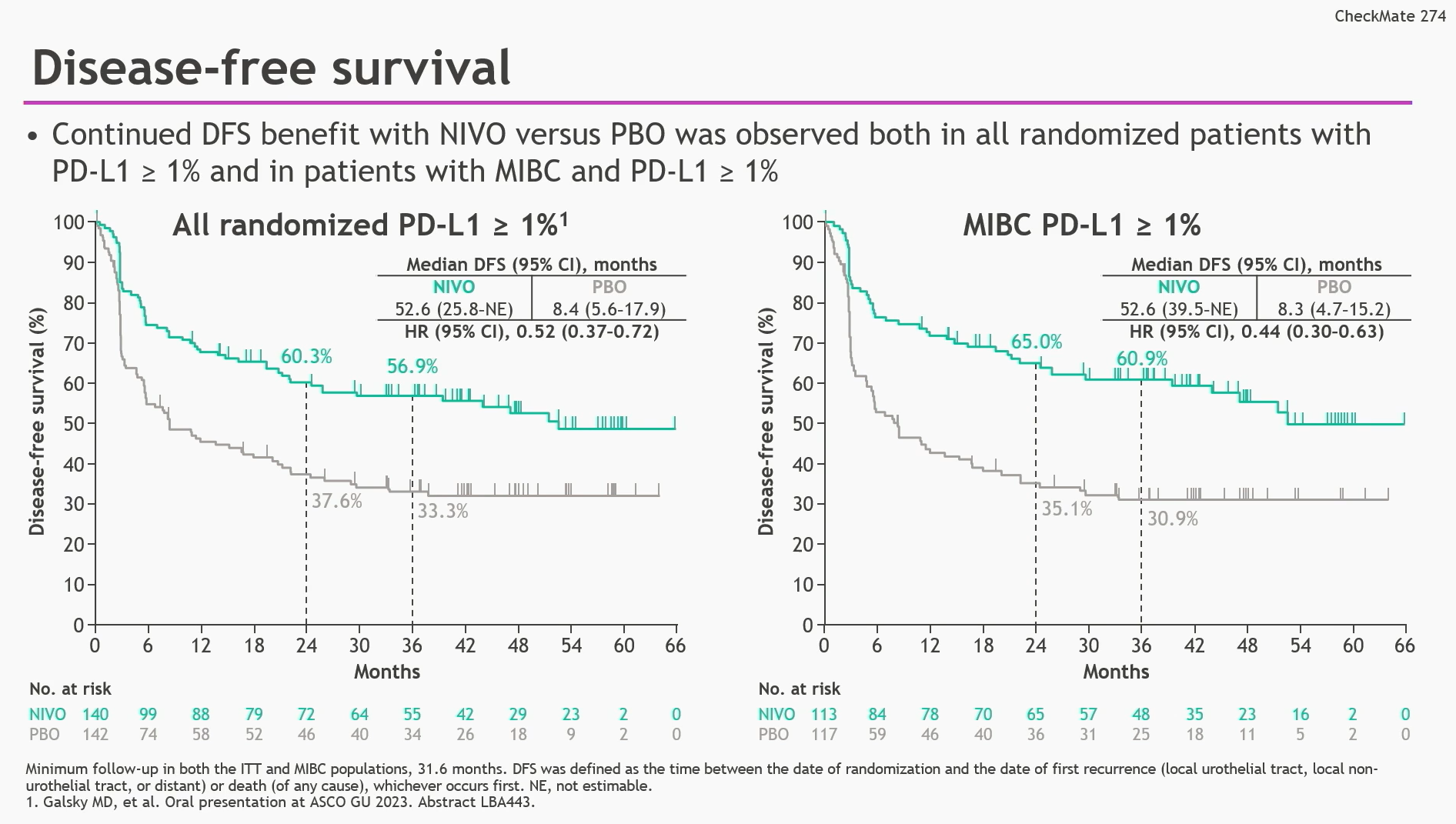
The EU Clinical Trials Register currently displaysĬlinical trials with a EudraCT protocol, of whichĪre clinical trials conducted with subjects less than 18 years old. Interventional clinical trials that were approved in the European Union (EU)/European Economic Area (EEA) under the Clinical Trials Directive 2001/20/ECĬlinical trials conducted outside the EU/EEA that are linked to European paediatric-medicine developmentĮU/EEA interventional clinical trials approved under or transitioned to the Clinical Trial Regulation 536/2014 are publicly accessible through theĬlinical Trials Information System (CTIS). Allows you to search for protocol and results information on: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
